Structure of glycoproteins
Glycoprotein carbohydrate chains are highly diverse. They are classified
into three groups:
- N-linked oligosaccharides ( N-glycans)
- O-Linked oligosaccharides ( O-glycans)
- Glycophosphatidylinositol ( GPI ) -membrane anchors
- The first sugar residue of N-glycans
is usually N-Acetylglucosamine (GlcNAc). It is linked to the amide
nitrogen of Asn in the protein. The target sequence for N-glycosylation
is Asn - X - Ser/Thr where X can be any amino acid residue except
Pro or Asp (Pro side chain would cause the steric hindrance and side chain
of Asp is negatively charged; it could make unfavour interactions with
negatively charged sugar residues). In some bacterial glycoproteins Asn
residue can be linked to Glc, GalNAc and L-Rha .N-glycans are divided into
three groups:
- High-mannose type
- Complex type
- Hybrid type
All of them have the common pentasaccharide core and they are
synthesized from a common precursor oligosaccharide. (see biosynthesis
of N-glycans )
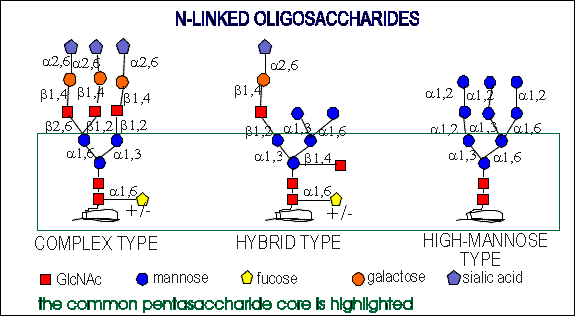
The N-linked oligosaccharides have a minimum of 5 sugar residues.
Complex type chains can be mono-, bi-, tri- (2,4 and 2,6 branched),
tetra-, and pentaantennary structures ( big variety). They can also contain
different amounts of sialic acid. High-mannose oligosaccharides can have
from 3 mannose residues to as many as 60 in protozoans and yeast.
N-linked glycoproteins exhibit many glycoforms. Cells synthesize
many variants of a given glycoprotein. Each variant (glycoform) differs
somewhat in the sequences, locations and numbers of its covalently bound
oligosaccharides.
- For example an N-linked oligosacchaeide chain of RNase B (RNase
A is carbohydrate-free) is microheterogenous : a sixth mannose residue
occurs at various positions on the core (GlcNAc)2 (Man)5
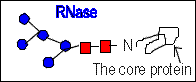
In that case the carbohydrate does not affect the conformation and substrate
specifity or catalytic properties of RNase A.
- The other glycoprotein - GM-CSF which promotes the developement,
activation and survival of granulocytes and macrophages, is variably glycosylated
at two N-linked and five O-linked sites.But in contrast to RNase B, the
degree of glycosylation of GM-CSF is important. It affects the lifetime
of this growth factor in the bloodstream (it increases with the level of
glycosylation).
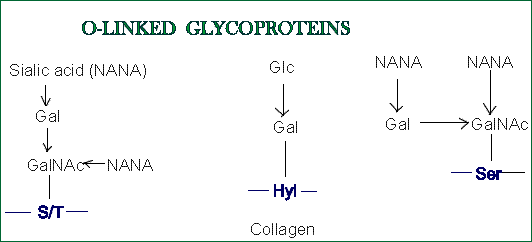
- In O-glycans oligosaccharides
are attached to a hydroxyl group of:
- Serine or threonine ; then the first sugar residue is usually
N-Acetylgalactosamine (GalNAc). Less commonly, galactose, mannose
or xylose form O-glycosidic bonds with Ser or Thr. Although most cytosolic
and nuclear proteins are not glycosylated, when they are, a single N-Acetylglucosamine
residue is linked to the Ser or Thr hydroxyl group. These exceptions include
some nuclear-pore complex proteins and some transcription factors.
- Hydroxylysine (Hyl); it is glycosylated by the attachment
of single Gal residue or glucosylgalactose disaccharide.
- Hydroxyproline (Hyp); arabinose residue is linked to
it.
Hyl and Hyp residues occur only in collagens.
O-linked oligosaccharides are generally short (1-4 sugar residues).
But for example o-glycans of ABO blood group antigens are longer.
The longest O-linked carbohydrate chains occur in proteoglycans.
They contain up to 1000 disaccharide units.
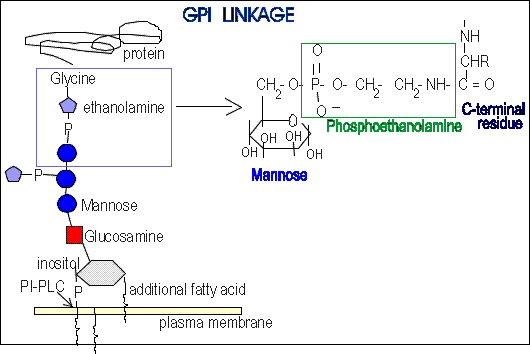
Glycophosphatidylinositol (GPI) membrane
anchors are attached to polypeptide chain through an amide bond
between mannose-6-phosphoethanolamine and the C-terminal carboxyl group
:
The example of GPI-linked protein can be Thy 1 which is the T-cell
marker. It interacts with proteoglycans in the extracellular matrix (ECM).
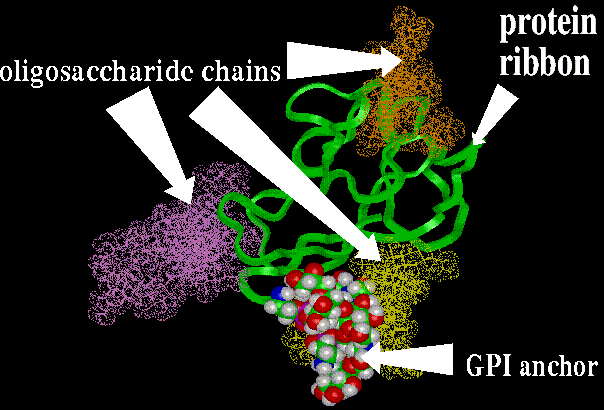
Other examples : acetylcholinesterase, intestinal and placental alkaline
phosphatase, adhesion molecules such as N-CAM from neural cells and LFA-3
(lymphocyte function associated antigen-3).
Go to :
Biosynthesis of glycoproteins
Contents
