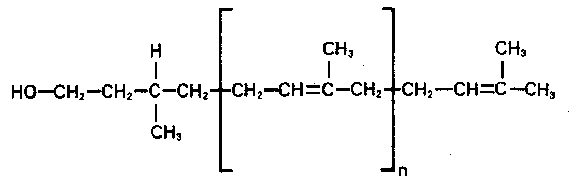Biosynthesis of N-linked glycoproteins
This process occurs in the ER and can be continued in the GA.
- First a lipid-linked oligosaccharide precursor is synthesized.
Its structure is the same in animals, plants and single-celled eucaryotes
:
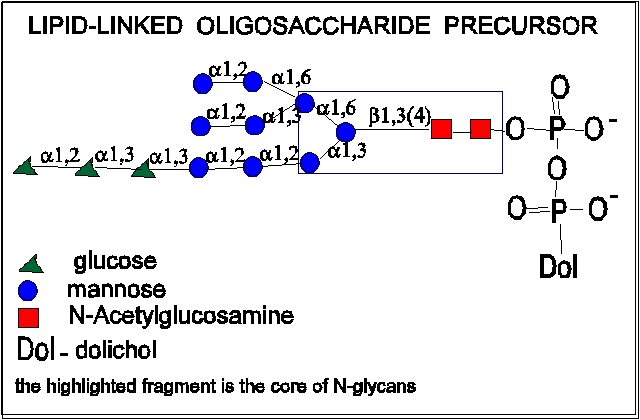
The precursor oligosaccharide is linked by a pyrophosphoryl group to dolichol.
It is a long (75-95 carbon atoms), highly hydrophobic polyisoprenoid lipid
:
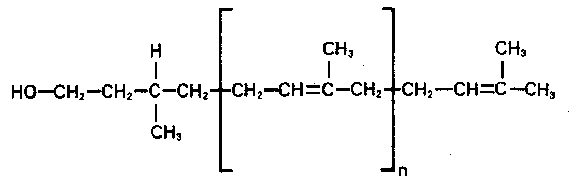
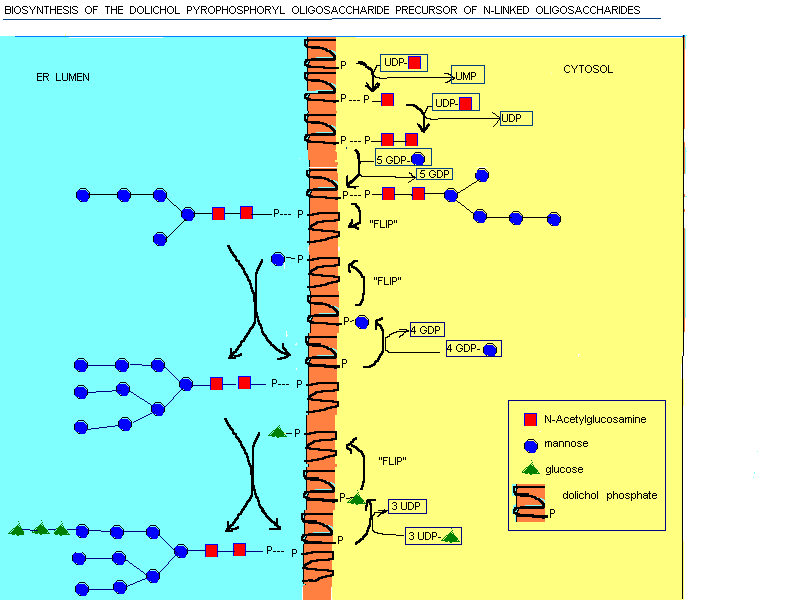
Dolichol is long enough to span the membrane 4 - 5 times. It is localized
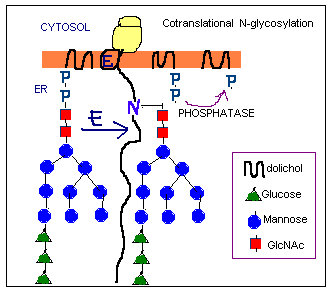
on the rough ER. The biosynthesis starts at the cytosolic face of the ER.
At the beginning 2 GlcNAc and 5 mannose residues are added one at time
to dolichol phosphate. Then the dolichol pyrophosphoryl oligosaccharide
is flipped to the luminal face. In the latter reactions each mannose or
glucose residue is transferred from nucleotide sugar to dolichol on the
cytosolic face of the ER. Then it is flipped and transferred to the growing
oligosaccharide. The carrier is then flipped back again to the cytosolic
face.
- The oligosaccharide precursor is transferred from the dolichol to an
Asn residue on the nascent polypeptide. Thus the glycosylation occurs
cotranslationally. The Asn residue must be in a sequence N-X-S/T
where X cannot be Pro or Asp.
The process is catalyzed by oligosaccharide protein transferase.
The enzyme is built up of three subunits. Two of them are ribophorins
- ER transmembrane proteins. Their cytosolic parts bind to the larger subunit
of the ribosome (they act as an anchor). The third subunit has the catalytic
activity.
Modifications of N-linked oligosaccharides
- ER : After an oligosaccharide chain have been added to the protein,
the three glucose and one mannose residues are removed by
three different enzymes in a fixed order. It occurs in the ER and is a
signal that the protein can be transported to the GA for further processing.
When the glycosylated protein is improperly folded the glucose residue
is again added to it. The oligosaccharide with glucose is recognized by
the membrane chaperon protein - calnexin. Glycoprotein bound to
calnexin cannot be exported to the Golgi (thus the quality of protein is
checked). When the conformation of the protein becomes proper the glucose
residue is removed and it is released from the calnexin.
After the processing in the ER the high-mannose type oligosaccharide
is formed.
 The high-mannose type glycoproteins
are transported (in membranous vesicles from the RER to the cis-cisternae
of the Golgi.
The high-mannose type glycoproteins
are transported (in membranous vesicles from the RER to the cis-cisternae
of the Golgi.
- GOLGI : Processing of glycoproteins in GA depends on their final
destination.
There are two main processing pathways :
For lysosomal enzymes
For non-lysosomal glycoproteins
Go to :
Biosynthesis of GPI-membrane anchors
Biosynthesis of glycoproteins
Contents