

|
To the newcomer, there may appear to be a somewhat confusing set of terms used to describe the different levels of structure which occur in proteins. Primary, secondary, supersecondary, tertiary, quaternary structure...folds, domains, motifs, modules, architecture, topology, mosaic proteins... to make matters worse, the precise meanings of some of these terms vary within the literature. An important point is that some of these terms (such as 'motif', 'domain') may be used either purely in the context of amino acid sequence, or in relation to three dimensional structure. There is of course an important relationship between sequence and structure (and a major goal of the biological sciences is to determine exactly what the details of this relationship are), so that in some cases the term 'domain' for example might apply both to the three-dimensional structure of a section of a polypeptide chain, as well as to its amino acid sequence (primary structure). It is therefore important to consider the context in which such terms are used.
To recapitulate, we have so far dealt with primary structure (amino acid sequence) and secondary structure. Secondary structure is defined by the phi and psi angles of the backbone atoms of the amino acid residues, and the hydrogen bonds between main chain atoms. In some cases these dihedral angles and patterns of hydrogen bonds are repeated throughout subsequences of several consecutive residues, giving rise to (most commonly alpha-)helices and beta-sheets. Alpha-helices and the beta-strands of sheets can therefore be described as 'units of secondary structure', as can turns and other non-repeating units.
Historically, the first hierarchical description of protein structure contained two more levels, in addition to primary and secondary (Linderström-Lang and Schellman, 1959). Tertiary structure concerns how the secondary structure units associate within a single polypeptide chain to give a three-dimensional structure. Quaternary structure describes how two or more polypeptide chains associate to form a native protein structure (but some proteins consist of a single chain).
These definitions are still valid, but more detail has since been added to the hierarchy. The introduction of the term "supersecondary structure" was necessary when it became clear that certain arrangements of two or three consecutive secondary structures (alpha-helices or beta-strands), are present in many different protein structures, even with completely different sequences.
Classic units of supersecondary structure include the alpha-alpha unit (two antiparallel alpha-helices joined by a 'hairpin' bend changing the chain direction by 180°); the beta-beta unit (two antiparallel strands connected by a hairpin); and the beta-alpha-beta unit (two parallel strands, separated by an alpha-helix antiparallel to them, with 2 hairpins separating the three secondary structures). Sometimes the term 'motif' is used to describe these supersecondary structures.
Other motifs of supersecondary structure have been described, such as the alpha-beta-beta and beta-beta-alpha units (Chothia, 1984).
Because supersecondary structure involves associations of secondary structures, supersecondary structure may logically be considered to be a subset of tertiary structure (see the definition above). On the other hand some literature may present supersecondary structure as a separate level in its own right.
It is important to realize that by no means all helices and strands in proteins belong to supersecondary structures. For example, proteins of the globin family consist of eight alpha-helices in contact; but the helices do not pack against other helices which are adjacent in the sequence, with the exception of the final two, which form an antiparallel helix-turn-helix motif. Again, the three- dimensional formation of these eight helices is the protein's tertiary structure.
Some relatively common combinations of the supersecondary structural motifs described above are observed in proteins. For example, there are a considerable number of proteins with a four-helix bundle, consisting of two alpha-alpha units connected by a loop. A common motif is the beta-alpha-beta-alpha-beta unit- alias the Rossman fold (effectively two consecutive beta-alpha-beta units sharing a strand). Arguably such units can be thought of as more complex supersecondary structural motifs.
These larger associations are also called (domain) folds. Some folds are considerably larger than the units described in the previous paragraph, consisting of several supersecondary structures, or secondary structures in other contexts. An example of the latter is the globin fold, six of whose helices cannot be described as a recognized supersecondary structure.
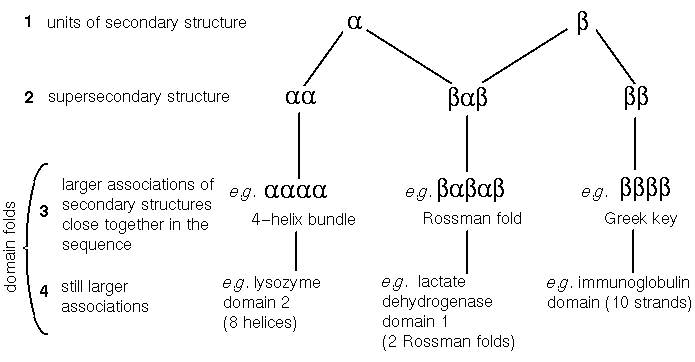
Figure 1. Levels 2, 3 and 4 come under the umbrella of 'tertiary structure', but tertiary structure can also describe how domains pack together. Possibly level 3 can be considered to constitute supersecondary structure as well as 2. Not all domain folds consist of motifs of supersecondary structure; for example the globin fold includes an alpha-alpha unit, but its remaining six helices do not comprise any 'classic' supersecondary motifs.
"Within a single subunit [polypeptide chain], contiguous portions of the polypeptide chain frequently fold into compact, local semiindependent units called domains." - Richardson, 1981
Domains may be considered to be connected units which are to varying extents independent in terms of their structure, function and folding behaviour. Each domain can be described by its fold. While some proteins consist of a single domain, such as the globins described previously, others consist of several or many. A number of globular protein chains consist of two or three domains appearing as 'lobes'. In other cases the domains may be of very different nature- for example some proteins located in cell membranes have a globular intracellular or extracellular domain distinct from that which spans the membrane.
Mosaic proteins are those which consist of many repeated copies of one or a few domains, all within one polypeptide chain. Many extracellular proteins are of this nature. The domains in question are termed modules and are sometimes relatively small. Note that this term is often applied to sequences, whose structures may not be known for certain.
Tertiary structure describes the association of units within domains, but tertiary structure also includes the way in which domains fit together. This should not be confused with quaternary structure, which concerns how separate polypeptide chains associate with each other. The domain can perhaps be considered the unit of tertiary structure (c.f. helices and sheets, the units of secondary structure).
A domain can be described as "all (or mostly)-alpha" (or just "alpha"); "all (or mostly)-beta" (or just "beta"); "alpha/beta", where it is rich in both helices and sheets combined in the classic supersecondary structure motifs (beta-alpha-beta) described earlier; or "alpha+beta" in which case it consists of helices and sheets which do not form such units. These four classes do not satisfactorily describe all folds however, such as small domains containing few helices or sheets; this is often the case with the modules described previously.
Again these terms might have slightly different meanings amongst the literature. For the purposes of this course, we will define them as follows.
Architecture will describe the orientations of secondary structures and the way they pack together. For example, a number of folds consist of two beta-sheets packing flat against each other at 90° forming a 'sandwich'. To describe this architecture, we need not be concerned about how the strands are related sequentially in the folded chain, and in the nature of the loops which connect them; these are considerations of topology.
Some proteins consist of a single polypeptide chain, such as myoglobin. The biologically functioning form of others is an aggregation of several copies of the same chain. For example, the quaternary structure of haemoglobin consists of four chains, each of which is similar to a myoglobin molecule. Other proteins exist of an aggregation of one or more copies of two different chains.
Note that some chains are formed by the proteolytic cleavage of an intact precursor chain. For example the active form of the digestive enzyme trypsin is formed from a single chain (the inactive form) which is cleaved into several shorter ones, without fundamentally altering the fold. Such cases should not be considered as a quaternary association of different chains.
Quaternary structure is the subject of a later chapter of the course.

|
Last updated 7th April '97