 Small Disulphide-rich Folds
Small Disulphide-rich Folds
![]() Index to Course Material
Index to Course Material
![]() Index to Section 10
Index to Section 10
![]() Alpha+Beta
Alpha+Beta
![]() References
References
Here we see examples of the main families of small disulphide-rich domains of
known structure. There are also `Small' domains without disulphides, which
could have been grouped here, but instead are grouped under all-alpha or all-beta
as appropriate (eg glucagon).
The Structural Classification of Proteins database (Alexey G. Murzin, Steven E. Brenner, Tim J.P. Hubbard, and Cyrus Chothia) currently lists thirty-eight categories of 'small' folds.
Also examine Jane Richardson's 'Protein Tourist 4' Kinemage, A Survey of Proteins in the 'Small Irregular' Category of Tertiary Structure.
![]()
Please note that the colour in the images below is purely artefactual, and merely serves to help in visualisation, but has no other significance. This form of representation does not actually show where the disulphide bonds are. To see this important feature, display the coordinate files served here with RasMol, and select from the menu Display: Wireframe, Sticks, Spacefill, or Ball & Stick, and Colour:CPK to see the yellow of the sulphur in the (usually buried) cysteines. Alternatively type the commands into the RasMol window: select sulphur; colour yellow; ssbonds 100 for example.
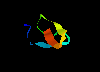 serine proteinase inhibitor - squash-type 2eti (39Kb)
[Bbk|BNL|ExP|Waw|Hal]
serine proteinase inhibitor - squash-type 2eti (39Kb)
[Bbk|BNL|ExP|Waw|Hal]
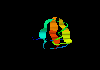 sea anemone toxin 1bds (66Kb)
[Bbk|BNL|ExP|Waw|Hal]
sea anemone toxin 1bds (66Kb)
[Bbk|BNL|ExP|Waw|Hal]
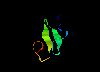 EGF-like domain 1ixa (48Kb)
[Bbk|BNL|ExP|Waw|Hal]
EGF-like domain 1ixa (48Kb)
[Bbk|BNL|ExP|Waw|Hal]
 insulin 4ins (117Kb)
[Bbk|BNL|ExP|Waw|Hal]
insulin 4ins (117Kb)
[Bbk|BNL|ExP|Waw|Hal]
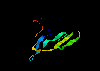 serine proteinase inhibitor - Bowman-Birk-type 1bbi (53Kb)
[Bbk|BNL|ExP|Waw|Hal]
serine proteinase inhibitor - Bowman-Birk-type 1bbi (53Kb)
[Bbk|BNL|ExP|Waw|Hal]
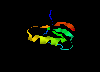 serine proteinase inhibitor - Kazal-type 2bus (80Kb)
[Bbk|BNL|ExP|Waw|Hal]
serine proteinase inhibitor - Kazal-type 2bus (80Kb)
[Bbk|BNL|ExP|Waw|Hal]
 serine proteinase inhibitor - Kunitz-type 1dtx (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
serine proteinase inhibitor - Kunitz-type 1dtx (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
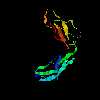 complement C-module domain (pair) 1hfh (147Kb)
[Bbk|BNL|ExP|Waw|Hal]
complement C-module domain (pair) 1hfh (147Kb)
[Bbk|BNL|ExP|Waw|Hal]
 snake toxin 2ctx (53Kb)
[Bbk|BNL|ExP|Waw|Hal]
snake toxin 2ctx (53Kb)
[Bbk|BNL|ExP|Waw|Hal]
 kringle domain 1pk4 (66Kb)
[Bbk|BNL|ExP|Waw|Hal]
kringle domain 1pk4 (66Kb)
[Bbk|BNL|ExP|Waw|Hal]
Last updated 19th Jun '96