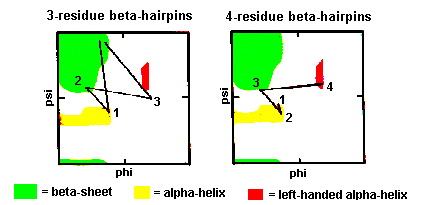
Normally the residues at the ends of the two beta-strands only make one hydrogen bond as shown below. The intervening three residues have distinct conformational preferences as shown in the Ramachandran plot. The first residue adopts the right-handed alpha-helical conformation and the second amino acid lies in the bridging region between between alpha-helix and beta-sheet. Glycine, asparagine or aspartate are frequently found at the last residue position as this adopts phi and psi angles close to the left-handed helical conformation. For RasMol-MIME, here is a pdb-file


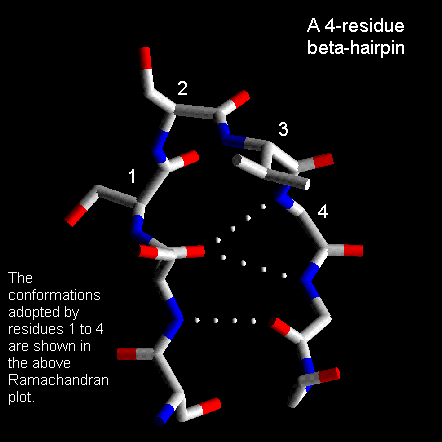
These are also quite common with the first two residues adopting the alpha-helical conformation. The third residue has phi and psi angles which lie in the bridging region between between alpha-helix and beta-sheet and the final residue adopts the left-handed alpha-helical conformation and is therefore usually Gly, Asp or Asn. Here is a coordinate structure file to view in RasMol.
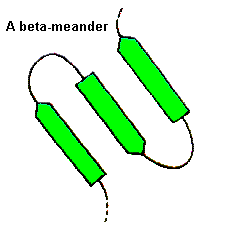
For these, a wide range of conformations is observed and the general term 'random coil' is sometimes used. Consecutive antiparallel beta-strands when linked by hairpins form a supersecondary structure known as the beta-meander.