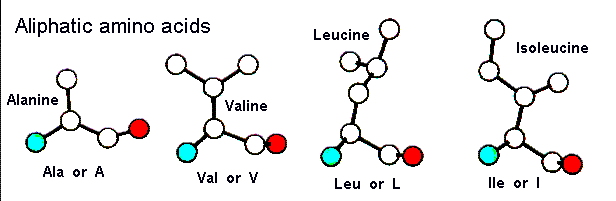
There are four amino acids in this class, as shown below. Their side chains consist of non-polar methyl- or methylene-groups. These amino acids are usually located on the interior of the protein as they are hydrophobic in nature. As can be seen, all of these side chains except for alanine are bifurcated. In the cases of Val and Ile the bifurcation is close to the main chain and can therefore restrict the conformation of the polypeptide by steric hindrance. In the below diagram the red and blue atoms represent polar main chain groups. Here is a pdb-file for RasMol.

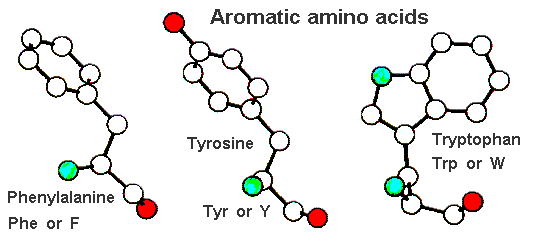
Of the three residues in this class (see below) only phenylalanine is entirely non-polar. Tyrosine's phenolic side chain has a hydroxyl substituent and tryptophan has a nitrogen atom in its indole ring sytem. These residues are nearly always found to be largely buried in the hydrophobic interior of a proteins as they are prdeominantly non-polar in nature. However, the polar atoms of tyrosine and tryptophan allow hydrogen bonding interactions to be made with other residues or even solvent molecules. Here is a pdb-file for RasMol.