A.Mills

Back to Protein Interactions Index
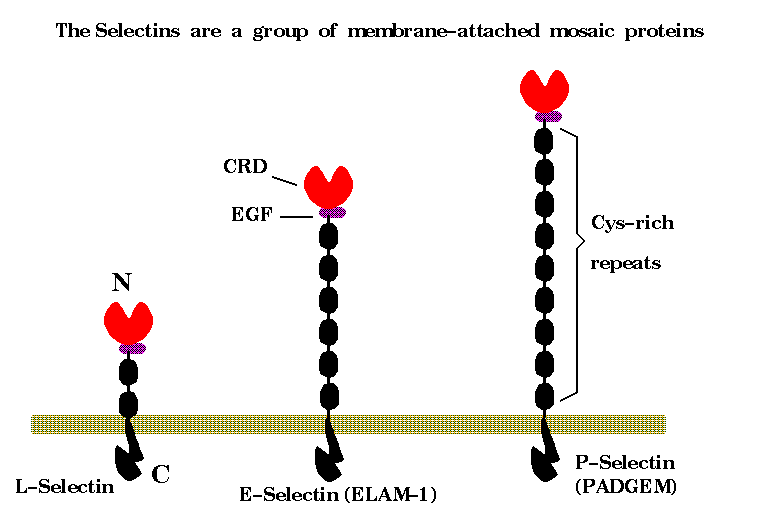
E-selectin, or ELAM-1 as it used to be called, is of considerable pharmaceutical interest as it is involved, along with ICAM-1, in recruiting leucocytes and macrophages into sites of inflammation. E-selectin and ICAM-1 are transiently expressed on endothelial cells lining the micro-vasculature when they are exposed to certain cytokines.
The counter-ligand to E-selectin is a complex oligosaccharide, known as sialyl Lewis-x, which is found on the surfaces of leucocytes (especially neutrophils), and it is the `stickiness' of endothelial cells which are expressing E-selectin which induces a temporary slowing-down, or rolloing action, as these cells of the lymphatic fluid float past.
The CRD domain is a member of the C-type animal lectins, a module found in a number of mosaic proteins of higher animals, whose sugar-binding capability is calcium-dependent. In 1992 Kurt Drickamer and Henderson solved the X-ray crystal structure of the homologous CRD from rat-mannose binding protein, and then later in complex with an oligosaccharide. The details of the calcium-dependent binding experiments were confirmed by mutagenesis. Around the same time, a large collaborative group of Genetech, GlycoMed, and Hoffman-LaRoche used mutagenesis and monoclonal antibodies to explore the details of the E-selectin/s-Lex interaction. Blocking antibodies and mutations were assessed by their effect on binding assays.
Crucial residues involved in the s-Lex binding were identified as Pro-46, Ser-47, Tyr-48, Arg-97, Lys-99, Lys-111, & Lys-113. These constitute a basic patch about 10 Angstroms from the calcium site.
Several efforts were made to model the E-selectin CRD structure, as well as its complex with sialyl-Lewis-x.
In 1994 the structure of the CRD of E-selectin was crystallographically determined, but, the structure of the complex is still (May'95) not available.
It is known that in mannose-binding protein, the 3'-OH and 4'-OH of mannose bind to the calcium ion and its liganding side-chains. It is suggested that in E-selectin, the 2'-OH & 3'-OH of fucose bind similarly. The s-Lex sialic acid carboxylate may then be attracted to the basic patch.
Here's Glenn Proctor's E-selectin modelling work, and here's a Back to the Top