 Part 1;
Part 1;
 Part 3
Part 3
Back to Protein Interactions Index
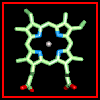 Click here for a diagram of haem.
Click here for a diagram of haem.
The haem group is situated between helices E and F, and is surrounded by non-polar residues except for the two carboxylate groups exposed at the protein surface (here is a diagram, and for two histidines. One of these (His 87 in alpha subunit, His 92 in beta), part of helix F, binds directly to the iron atom of the haem group (the NE2 atom of the His side chain occupies one of the six coordination positions of the iron). This His is in each subunit called the proximal histidine. The distal histidine occurs in helix E (His 58 in alpha subunit, His 63 in beta). This is near to the opposite coordination position, but does not occupy it; this coordination site is occupied by oxygen in oxyhaemoglobin.
 Here are orthogonal views of
(oxy)haemoglobin illustrating the positions of the proximal and distal
histidines in relation to the haem.
Here are orthogonal views of
(oxy)haemoglobin illustrating the positions of the proximal and distal
histidines in relation to the haem.
 The orientation of the four
subunits in the tetrameric haemoglobin molecule is indicated in this diagram.
The orientation of the four
subunits in the tetrameric haemoglobin molecule is indicated in this diagram.
Swiss-3D prot provide this diagram.
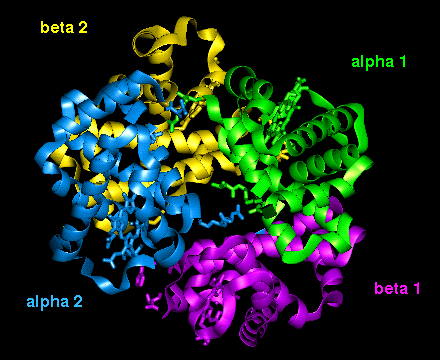 This diagram illustrates (1) and
(2) of the above.
This diagram illustrates (1) and
(2) of the above.
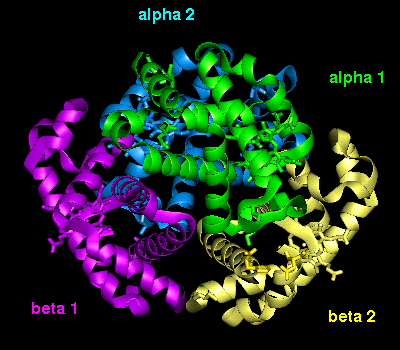 This diagram illustrates (3) and
(4).
This diagram illustrates (3) and
(4).
These interactions may also be examined in this structure which includes side chains only for the residues mentioned above .
Here is the complete structure of deoxyhaemoglobin.
The haem group of deoxyhaemoglobin is domed rather than planar. This relates to the ionic radius of the iron, which is in a high-spin Fe(II) state. The iron is too large (radius 2.06Å) to fit in the ring of nitrogens with which it coordinates; it is 0.6Å out of the plane of the ring, which is therefore distorted.
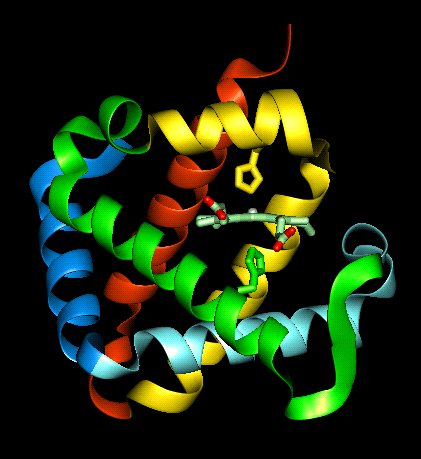
 Close-up
Close-up
 and a
perpendicular view
and a
perpendicular view
In contrast to deoxyhaemoglobin, the haem group is planar and the iron ion lies in the plane of the ring, as it is in a low-spin Fe(II) state with a smaller radius (1.98Å). All six coordination positions of the ion are occupied: the bound oxygen molecule accounts for the sixth.
Examine the structure of oxyhaemoglobin (tetramer provided by Brookhaven).
Perpendicular view:
 Allosteric Mechanism
Allosteric Mechanism 
Back to Main PPS Index
J. Walshaw