 Super Secondary Structure
Super Secondary Structure
![]() Index to Course Material
Index to Course Material
![]() Index to Section 9
Index to Section 9
![]() Index to Super Secondary Structure
Index to Super Secondary Structure
![]() Next Page
Next Page
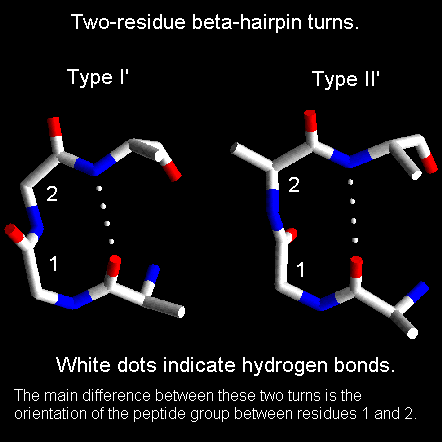
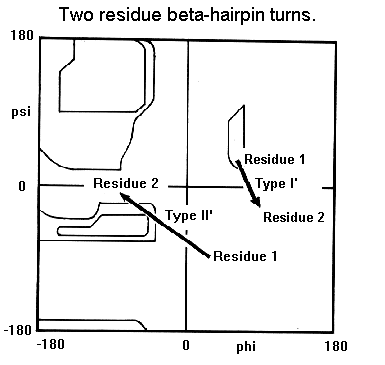
Beta-hairpins are one of the simplest supersecondary structures and are widespread in globular proteins. They occur as the short loop regions between antiparallel hydrogen bonded beta-strands. In general a reverse turn (or beta-turn, as it they are sometimes called) is any region of a protein where there is a hydrogen bond involving the carbonyl of residue i and the NH group of residue i+3. An alternative definition states that the alpha-carbons of residues i and i+3 must be within 7.0 Angstroms. The structures of reverse turns are outlined in the section on peptide geometry. In this section we will concentrate on those turns which occur between consecutive beta-strands, known as beta-hairpins. Sibanda and Thornton have devised a system for classifying beta-hairpins which is based on two conventions for defining loop regions. In this section we will not go into such details as the objective is indicate the most commonly observed hairpin loop structures.
Beta-hairpin loops adopt specific conformations which depend on their lengths and sequences. Sibanda and Thornton have shown that 70% of beta-hairpins are less than 7 residues in length with the two-residue turns forming the most noticeable component. These two-residue beta-hairpins all adopt one of the classical reverse turn conformations with an obvious preference for types I' and II'. Type I 2-residue hairpins also occur but with lower abundance. This contrasts with reverse turns where types I and II tend to dominate. In beta-hairpins the type I' turn has the correct twist to match the twist of the beta-sheet and modelling studies indicated that if either type I or type II turns were to connect the antiparallel beta-strands, they would diverge within a short distance from the turn.
Last updated 31st May '96