 Alpha/Beta Doubly-Wound Folds
Alpha/Beta Doubly-Wound Folds
![]() Index to Course Material
Index to Course Material
![]() Index to Section 10
Index to Section 10
![]() Alpha/Beta Folds
Alpha/Beta Folds
![]() Alpha+Beta Folds
Alpha+Beta Folds
The doubly wound alpha/beta topology was described on the
previous page. Some variations of the classic
doubly wound fold are illustrated here. In the topology diagrams below, the
parts of the chain linking the beta-strands are red, and are thick when they
include an alpha-helix. In the RasMol scripts, non-alpha-helical linking regions
are shown in pink. Some of the scripts highlight in blue where the fold begins.

![]() flavodoxin, 1flv (127Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
flavodoxin, 1flv (127Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT

![]() subtilisin, 1sbt (172Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
subtilisin, 1sbt (172Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT

![]() arabinose-binding protein, domain 1, 5abp (217Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
arabinose-binding protein, domain 1, 5abp (217Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT

![]() glyceraldehyde phosphate dehydrogenase, 3gpd (443Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
glyceraldehyde phosphate dehydrogenase, 3gpd (443Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT

![]() dihydrofolate reductase, 3dfr (157Kb)
[Bbk|BNL|ExP|Waw|Hal]
dihydrofolate reductase, 3dfr (157Kb)
[Bbk|BNL|ExP|Waw|Hal]

![]() adenylate kinase, 3adk (135Kb)
[Bbk|BNL|ExP|Waw|Hal]
adenylate kinase, 3adk (135Kb)
[Bbk|BNL|ExP|Waw|Hal]

![]() rhodanese domain 1, 1rhd (202Kb)
[Bbk|BNL|ExP|Waw|Hal]
rhodanese domain 1, 1rhd (202Kb)
[Bbk|BNL|ExP|Waw|Hal]

![]() glutathione reductase, 1gra (366Kb)
[Bbk|BNL|ExP|Waw|Hal]
glutathione reductase, 1gra (366Kb)
[Bbk|BNL|ExP|Waw|Hal]

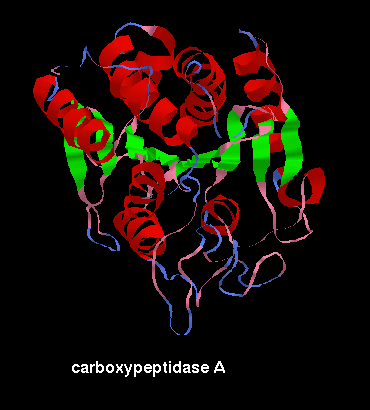
![]() carboxypeptidase, 2ctb (Kb)
[Bbk|BNL|ExP|Waw|Hal]
This fold varies considerably
from a classic doubly-wound variation in order to achieve the arrangement of
sheet (mixed parallel and antiparallel in this case).
carboxypeptidase, 2ctb (Kb)
[Bbk|BNL|ExP|Waw|Hal]
This fold varies considerably
from a classic doubly-wound variation in order to achieve the arrangement of
sheet (mixed parallel and antiparallel in this case).
Last updated 17th Jun '96