Dr Tracey Barrett
Our main interests lie in the area of DNA repair with particular emphasis on proteins
involved in base excision repair, eukaryotic homologous recombination and the processing
of general DNA damage.
Other interest include proteins that are involved in tumour susceptibility together
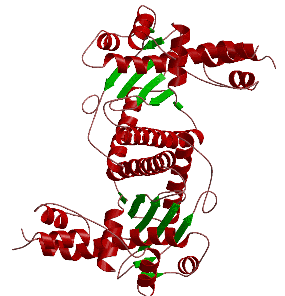
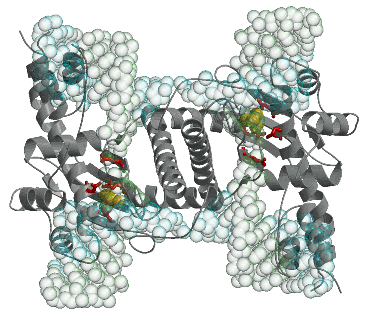
with their interacting partners. The crystal structure of the first eukaryotic Holliday
junction resolvase (Ydc2) solved within the group is shown below as a cartoon (left)
and an electrostatic potential surface (right).
A model for a Ydc2-junction complex has also been constucted.
Positions held:
1998-2003 BBSRC David Phillips Fellow
2003- Lecturer
Current group members are:
Dr Simona Ceschini Postdoctoral Fellow
Dr James Ryan
Postdoctoral Fellow
Mr Juan Sanchez-Weatherby Mres student
Relevant Publications:
Tracey E. Barrett, Renos Savva, George Panayotou, Tom Barlow, Tom A Brown, Josef Jiricny and
Laurence H. Pearl. Crystal structure of a G:T/U Mismatch-Specific DNA Glycosylase: Mismatch
recognition by complementary-strand interactions.Cell, (1998),92, 117-129.
Tracey E. Barrett, Renos Savva, George Panayotou, Tom Barlow, Tom A Brown, Josef Jiricny and
Laurence H. Pearl. Structure of a DNA base-excision product resembling a cisplatin inter-strand
adduct.
Nature Structural Biology, (1998), 5, 697-701.
Tracey E. Barrett, Orlando D. Scharer, Renos Savva, Tom Brown, Josef Jiricny, Gregory Verdine,
and Laurence H. Pearl. Crystal structure of a thwarted mismatch glycosylase DNA repair complex.
Embo. J, (1999), 18, 6599-6609.
Simona Ceschini, Anthony Keeley, S.B.Mark McAlister, Mark Oram, John Phelan, Laurence H. Pearl,
Irina R. Tsaneva, Tracey E. Barrett. Crystal structure of the fission yeast mitochondrial
Holliday junction resolvase Ydc2. Embo. J, (2001), 20, 6601-6611.
Magnar Bjørås, Erling Seeberg, Luisa Luna, Laurence H. Pearl and Tracey E. Barrett.
Reciprocal "Flipping" underlies substrate Recognition and catalytic activation by the
human 8-oxo-guanine DNA glycosylase. J.Mol.Biol. (2002), 317, 171-177.