

|
|
Insulin
Monomers, Dimers and Hexamers
Insulin exists in solution (depending on concentration and pH) as monomers or dimers and in rhombohedral crystals as hexamers. The oligomerization of insulin results in small variations in side-chain and main-chain structure. This is true in the soluble dimer and hexameric crystal forms. These changes depend on the oligomersization state and reflect the constraints that the different quaternary structures impose on the tertiary structures. It is interesting to note that though the tertiary structure is, to some degree, governed by the quaternary structure, the insulin fold and the activity of the molecule are preserved.
The complete amino acid sequence of porcine insulin was determined by Sanger and co- workers in 1955 (Brown et al., 1955). Insulin has a molecular weight of 5808 daltons and contains 51 amino acids, arranged in two chains (an acidic A chain of 21 residues and a basic B chain of 30 residues) which are cross-linked by two disulphide bridges. A third disulphide bridge links two parts of the shorter A chain. (See Ryle et al.,1955).
Although insulin was first crystallized by Abel and co-workers in 1925, it was not until 1934 that Scott discovered that the rhombohedral crystals were a zinc-insulin complex. Together with Fischer (Scott and Fischer 1935), Scott showed that the zinc might be replaced by other divalent ions. In time, this opened up the possiblity that the crystal structure might be solved by isomorphous replacement. An initial x-ray analysis of porcine zinc insulin at 2.8 Å was completed in 1969 by Hodgkin and co-workers (Adams et al. ;1969, Blundell et al. ;1971) and at 2.5Å by the Peking Insulin Structure Research Group in 1971. 2-zinc insulin has since been analysed at 1.9 Å resolution (Blundell et al. ,1972a), at 1.8Å resolution (Peking Insulin Structure Research Group, 1974) and more recently refined to 1.5 Å (Sakabe et al.;1977; Dodson et al., 1980) and to 1.1 Å resolution (Sakabe et al., 1981). In the rhombohedral crystal, the unit cell contains six molecules of insulin. The hexamer is assembled from three equivalent dimers which are related by a 3-fold axis. Two zinc ions lie 17 Å apart on the 3-fold axis, each of them co-ordinated by B10 His from each of the 3 dimers and 3 water molecules (see figure below).
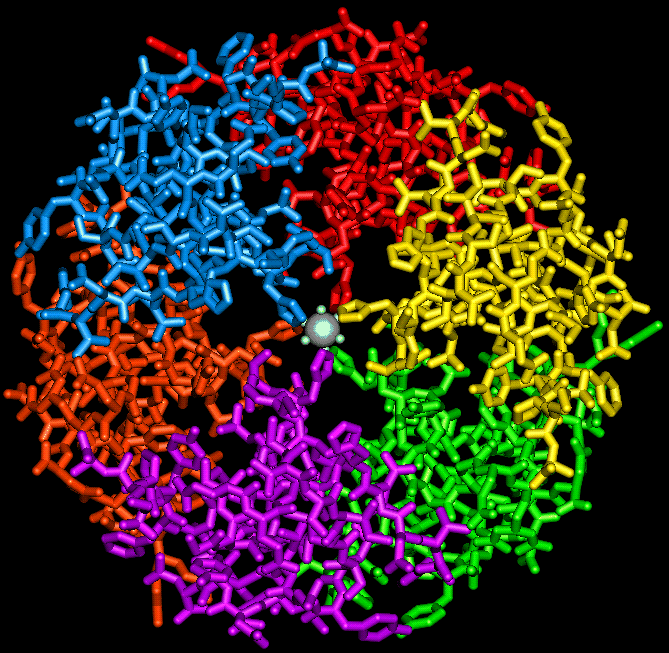
The insulin hexamer viewed down the 3-fold axis, indicating the position
of the zinc ions and the three water molecules which coordinate with each
of them.
A view of the hexamer perpendicular to the 3-fold axis
![]() Here is the
pdb file (203Kb). It contains no solvent
molecules other than those waters which coordinate with each zinc ion.
SCRIPT 1 renders the molecule as in the diagrams
above, displaying each insulin monomer (one A + one B chain) in a different
colour. Keep this RasMol window open for further scripts below.
Here is the
pdb file (203Kb). It contains no solvent
molecules other than those waters which coordinate with each zinc ion.
SCRIPT 1 renders the molecule as in the diagrams
above, displaying each insulin monomer (one A + one B chain) in a different
colour. Keep this RasMol window open for further scripts below.
Here are spacefilling and ribbon ribbon renditions of the hexamer.
The insulin monomer is a compact globular structure with a hydrophobic core. Although the surface residues are primarily polar, there are two hydrophobic surfaces on each side of the molecule which are buried during the formation of dimers and 2-zinc hexamers. In the insulin fold, the A chain is a compact unit around which the B chain is wrapped.
The A chain consists of two anti-parallel stretches of imperfect alpha helices (A2 Ile - A8 Thr and A13 Leu - A19 Tyr) which are joined by a turn from A9 Ser to A12 Ser, stabilized by the A6-A11 disulphide. The A chain lies in a plane in which the N and C terminii are brought to the same side, bringing A2 Ile and A19 Tyr into van der Waals contact (see diagram).
The B chain consists of an alpha-helix (B9 Ser - B19 Cys) from which both N and C terminii residues extend. The glycine residues at B20 and B23 allow the chain to fold back on itself in an approximate V- shape, and this brings the C terminal residues B24 Phe and B26 Tyr into van der waals contact with B15 Leu and B11 Leu of the alpha-helix (see diagram).
The insulin fold is formed when interchain disulphides at A7 and A20 form interchain disulphides with the B chain cysteines at B7 and B19 respectively. The (A7-B7) disulphide is fully exposed on the surface of the molecule, whereas the (A20-B19) disulphide is part of the hydrophobic core (see next diagram). Burial of the intrachain (A6 - A11) disulphide and the non-polar side chains of A16 Leu, B11 Leu, B15 Leu, A2 Ile and B24 Phe provides the hydrophobic interior stabilising the fold. The N termini residues of the B chain are folded across and run anti-parallel to the turn in the A chain, giving rise to hydrogen bonding between A11 Leu and B4 Gln, A7 Cys and B5 His, and between A19 Tyr and B25 Phe. Further stability is also provided by a salt bridge between the polypeptide chains at A11 Cys and B4 Gln, between the A7 carbonyl oxygen and the B5 His side chain, and between the A19 carbonyl oxygen and the B25 back-bone nitrogen. Further stability is also provided by a salt bridge between B29 Lys and A4 Glu and between the positively charged B22 Arg side chain and the negatively charged A21 terminal carboxyl group (in molecule 1 only).
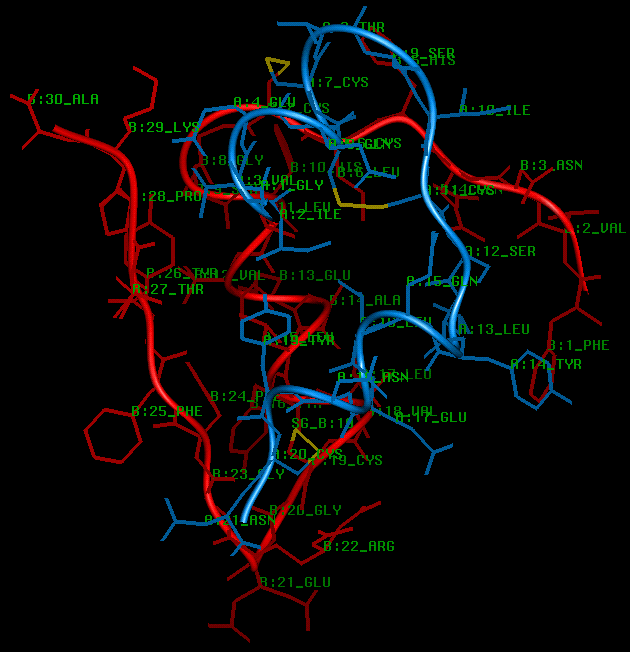 SCRIPT 4
SCRIPT 4
The insulin monomer viewed perpendicular to the 3-fold axis. The 2 interchain
and 1 intrachain disulphides are shown in yellow.
In solution at neutral pH and at physiological concentrations (about 1 ng/ml) insulin exists as a monomer (Frank et al.,1972), and it is the monomer which is the active form of the hormone (Cahill, 1971). At higher concentrations at acid or neutral pH (in the absence of zinc) the insulin monomer self-associates to form dimers and (in the presence of zinc) hexamers (Jeffrey and Coates, 1966; Carpenter, 1966). Studies of insulin analogues demonstrate how activity depends on the integrity of the insulin fold, and also allow mapping of the interactive residues on the surface of the molecule. The central residues responsible for hexamer formation include B10 His, which binds zinc ions, and B14 Ala, B17 Leu, B20 Gly and A 13 Leu, which are involved in close-packed hydrophobic interactions. The ability to form dimers is mediated by hydrophobic interactions involving B8 Gly, B9 Ser, B12 Val, B13 Glu, B16 Tyr, B24 Phe, B25 Phe, B26 Tyr, B27 Thr and B28 Pro (Baker et al. , 1988; Brange et al., 1990). The B chain of one monomer packs against the B chain of the second monomer and further stability is provided by hydrogen bonding between the antiparallel beta-strands B24 - B26 of each molecule. The packing arrangement in the dimer results in perturbations of side-chain and main-chain structure (Chothia et al., 1983), such that the monomers differ slightly in conformation. The B25 Phe side-chain of molecule 2 is squeezed from its pocket in molecule 1 and appears on the monomer-monomer interaction surfaces, as indicated below.
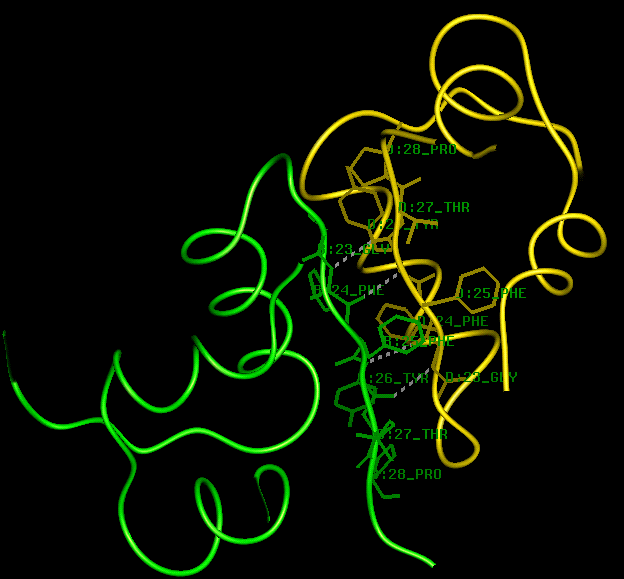
To examine the porcine insulin dimer (2x[A+B]), either continue using the same hexamer PDB structure file and type
select *a,*b,*c,*d
then select a rendering option from the RasMol menu. Alternatively, download the crystal structure from which this hexamer was generated: 4ins (117Kb) [Bbk|BNL|ExP|Waw|Hal].
The conformation of the insulin monomer varies from one crystal form to another (for review see Derewenda et al. , 1990). Although the insulin-fold is always retained, the major secondary structure elements are capable of individual rigid body movements which are accomodated by alterations in side-chain positions (Lesk 1991). The variety of insulin quaternary structures have been the subject of much discussion, but the differences are mainly localised to the N- and C- terminii of the B-chain (Dodson et al. , 1980). The most extreme variation is in the N-terminus of the B chain which may exist either in an extended conformation (T state) or as an extension of the B chain helix (R state; Kaarsholm et al. , 1989; Kruger et al., 1990; see below).
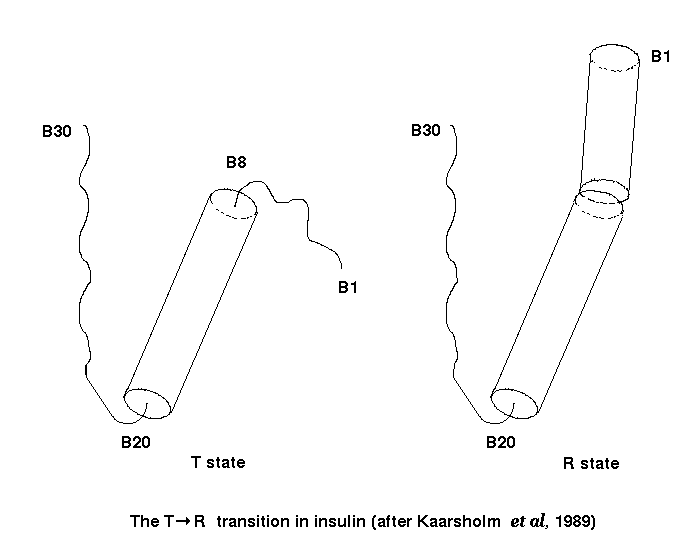
That these conformational variations are not simply due to crystal packing forces is demonstrated by molecular dynamics simulations performed in vacuo which highlight relative movement of the helices and flexibility of the chain termini (Kruger et al. , 1987; Caves et al., 1990). The T to R transition can be reproduced reversibly in the restricting environment of the crystal lattice, demonstrating the flexibility inherent in the insulin molecule (Bentley et al. , 1978). The T to R transition does not appear to be important in receptor binding since gamma-ethylnorleucine B6 insulin, which has limited ability to undergo the transition, still binds the insulin receptor with high affinity (Nakagawa and Tager, 1991).
Since the initial X-ray analysis of insulin (Adams et al., 1969), there have been a series of crystallographic studies on monomeric, dimeric and hexameric insulins in several forms. Some of these insulins have been variants and others have been chemically synthesized, modified or mutated by recombinant DNA techniques.
Although there are variations in the molecule's conformation in the different crystals and between species, the insulin fold is essentially maintained. As the native insulin monomer will always form multimers at the concentrations necessary for crystal growth, a crystal structure analysis of the native monomeric molecule is impossible. However, despentapeptide insulin which is unable to form the hydrogen bonded beta-sheet between the monomers , does crystallize as a monomer (although there are still 2 molecules in the unit cell) and gives monoclinic crystals (Bi et al., 1983,1984). In the absence of metal ions native insulin crystallizes as a dimer. In some crystals the two-fold axis is exact and crystallographic (e.g. neutral pH cubic crystals; Dodson et al., 1978, and neutral pH tetragonal crystals; Cutfield et al., 1979) and the insulin monomer occupies the asymmetric unit. In other crystals the two-fold axis is approximate and non-crystallographic (e.g. low pH orthorhombic human insulin crystals; Derewenda et al., 1989a). The two B25 Phe side-chains respond to the symmetry of the dimer ; in a symmetrical dimer both are directed away from the two-fold axis, while in the asymmetrical dimer one side turns away and contacts its partner across the approximate axis. The conformation of the insulin molecule which persists in nearly all crystal forms and probably also in the circulation is referred to as molecule 2 (molecule 1, Chinese convention), its partner in the asymmetric dimer is referred to as molecule 1 (molecule 2, Chinese convention). In the dimerization of two insulin monomers the two B13 Glu side chains are directed away from each other and solvated, minimizing their repulsion (Badger et al., 1991). In hagfish insulin, which has an Asn residue at B13, the two molecules of the dimer are identical and resemble molecule 2 of the porcine dimer (Cutfield et al., 1974; Peterson et al., 1975).
In the presence of zinc ions, three identical insulin dimers assemble into a hexamer in which each of two zinc ions lies on a three-fold axis and resides in a distorted octahedral ligand field co-ordinated by three B10 His imidazolyl groups and by three water molecules. The hexamer is stabilized by hydrophobic interactions between B1 Phe, B14 Ala, B17 Leu, A13 Leu and B18 Val residues. The local two-fold axis within each dimer is perpendicular (or approximately so) and intersects the three-fold axis. X-ray crystallographic studies of porcine 2 zinc insulin (Blundell et al.,1972a) showed that the hexamer is torus shaped (50 Å in diameter by 35 Å high) and that the two zinc ions lie 17 Å apart on either side of the hexamer centre, on the three-fold axis which traverses the central cavity. In the porcine 2Zn insulin crystal (T6) the dimer is the asymmetric unit (Baker et al., 1988).
The insulin hexamer is toroidal, highly stable and has a polar surface, making it ideal for crystal formation. In pancreatic beta-cells (which have a high content of zinc), mature secretory granules contain insulin which forms a dense array of 50 Å units with packing similar to that found in the crystalline hexamer (Greider et al., 1969), and it has been proposed that the 2 Zn hexamer is the storage form of insulin (Blundell et al., 1972a). Crystallization within the storage granule seems to serve two purposes, firstly to act as an efficient store of insulin and secondly to protect insulin molecules from proteolytic processing.
It has been shown that anions induce the T to R transition at residues B1-B8 of the insulin molecule (Bentley et al., 1975; Renscheidt et al., 1984; Kaarlsholm et al, 1989). Transformation efficiency follows the Hoffmeister lyotropic series (Harding et al, 1966, De Graaf et al., 1981) and some residues move by as much as 20-25 Å (Smith et al., 1984 ; Derewenda et al., 1989). Chloride ion concentrations above 6% have a dramatic effect on the insulin hexamer, inducing the T to R transition in three molecules (molecule 1), which now form a three-fold symmetrical trimer, out of the six, and causing a cleft to appear in these molecules due to movement of the (A7-B7) disulphide (molecule 2 remains in the 2 Zn insulin conformation). The movement apart of the A-chain and the B-chain C-terminii breaks the hydrogen bond between A19 Tyr and B25 Phe and exposes the hydrophobic core residues. The approximate two- fold symmetry between the dimers in the 2Zn hexamer is lost, however the axial symmetry relating the monomers in the dimer is preserved. At one face of the hexamer, a single octahedral zinc site coordinated by three B10 His residues (of molecule 2) and three water molecules is retained. At the other face of the hexamer the helical conformation of the B1-B8 segments of the other three B-chains brings the B5 his (from molecule 1) and B10 His (from molecule 1) rings from adjacent dimers together, creating two types of additional zinc sites. At these new faces, the B10 His side chain can exist in two distinct orientations. In one orientation the symmetrically related B10 His side chains and a chloride ion (or water molecule) coordinate a zinc ion on the 3-fold axis. Alternatively, the B10 His side chains may be rotated about the C-alpha-C-beta bond to bring them into close proximity with the imidazole ring of B5 His from the adjacent dimer, creating three identical off-axial sites. Each off-axial zinc ion is coordinated by two chloride ions (or water molecules) and two histidyls (B5 His and B10 His). The off-axial sites are not always fully occupied (Smith et al., 1984) but at high zinc concentrations 4 Zn hexamers can be formed (Schlichtkrull, 1958). In the 4 Zn (T3,R3) porcine and human insulin crystals, the dimer is still the asymmetric unit (Bentley et al., 1976, Smith et al., 1984). The 2 Zn to 4 Zn crystal transformation can be elicited by soaking the crystalline 2 Zn hexamer in high concentrations of anions without distorting the crystal lattice. If the chloride ion concentration is reduced below 6%, the 4 Zn (T3,R3) crystal reverts back to the 2 Zn (T6) structure (Bentley et al., 1978). Alternatively, the 4 Zn hexamer can be crystallised directly.
Phenol has also been shown to induce the 2Zn to 4Zn transformation in solution (Wollmer et al., 1987), but 2Zn insulin crystals do not survive soaking in phenol without cracking. However, crystallization in the presence of phenol at levels of 0.5%, causes the B-chain helices of all insulin molecules to extend from B1 to B20, giving the dimers approximate two-fold symmetry. Each phenol binds in a hydrophobic cavity created by the packing of the B1 to B8 helix against the A chain of the three-fold related dimer. More specifically, the phenol hydroxyl group hydrogen bonds to the (A6-A11) cysteine bridge and the aromatic ring makes van der Waals contact with B5 His of the adjacent dimer, blocking off the axial zinc-binding sites. The resulting 2 Zn (R6) crystals are monoclinic with a hexamer in the asymmetric unit (Derewenda et al., 1989a). Phenol is commonly used as a preservative in commercial insulin preparations at a level of approximately 0.2%, and interestingly such levels can occasionally convert rhombohedral crystals to the monoclinic form (Brange, 1987).
When insulin is mixed with protamine in the presence of zinc, phenol or mcresol, a precipitate is formed which can be converted to tetragonal crystals (Krayenbuhl and Rosenberg, 1946). Chemical analysis indicates the presence of two zinc ions and 22 phenol molecules per hexamer and one protamine per 8.5 insulin monomers. X-ray studies reveal that the hexamer in these crystals is symmetric, with three two-fold axes as is the case for monoclinic crystals (Derewenda et al., 1989b).
The reluctance of native insulin to form hexamers in the absence of zinc ions suggests that there are unfavourable interactions which are overcome by the zinc coordination to B10 His. The repulsive potential of the six B13 Glu carboxylate groups at the centre of the insulin hexamer has been predicted to limit the formation of zinc-free insulin hexamers at neutral pH (Blundell et al.,1972 ; Coffman and Dunn, 1988 ; Baker et al., 1988). In the 2 Zn hexamer the B13 Glu side chains are arranged as three hydrogen-bonded pairs, an arrangement which reduces their electrostatic repulsion at the expense of the energy required to protonate half of the carboxylates. Solution studies including osmometry (Roy et al., 1989; Hansen 1991), volume exclusion chromatography and circular dichroism studies (Wollmer et al., 1989) have established the existence of a stable metal-free hexamer when B13 Glu is mutated to Gln. X-ray analysis of the zinc-free B13 Gln insulin hexamer reveals that the glutamine side-chains are entirely hydrated and make no hydrogen-bonding contacts (Bentley et al., 1992). Surprisingly the organisation of the zinc-free hexamer is T3,R3 in the absence of any anions ; either the B13 Gln mutation favours the R state or the sulphate ion stabilizes the B1 - B8 helix (ammonium sulphate is not used for 2Zn hexamer growth as a change in pH is sufficient to cause supersaturation). Inspection of known insulin sequences shows that a carboxylic acid residue (Glu or Asp) at B13 is almost universal in all zinc binding insulins (Hallden et al., 1986), the exception being the unusual Atlantic Hagfish insulin where B13 is an asparagine and which is thought not to form hexamers (Peterson et al., 1975 ; Cutfield et al., 1979), suggesting that it has important structural or functional properties. It has been proposed that the function of B13 acidic groups is to drive apart the hexamer as soon as the stabilizing zinc ions diffuse away upon expulsion of the hexamer from the beta-cell (Bentley et al., 1992). However, B13 Glu is conserved in Guinea Pig insulin which is known not to hexamerise (Zimmerman and Yip, 1974), suggesting that its role may not be limited to hexamer assembly. Commensurate with this is the 80% fall in potency of B13 Gln insulin in the free fat cell assay (Marki et al., 1979) and a fall of 60% in the mouse blood glucose assay (Markussen et al., 1987a).
The work of Schlichtkrull (1956) on the stoichiometries of divalent metal ion binding in crystalline rhombohedral insulin hexamers gave the first indication for the presence of a third, high affinity metal binding site within the insulin hexamer. During the initial X-ray structure determination of the 2 Zn hexamer, isomorphous replacement studies located the extra metal binding site to the centre of the hexamer (Adams et al., 1967) and further studies showed that the B13 Glu residues specifically bound metal (Blundell et al., 1972; Emdin et al., 1980). NMR experiments involving metal ion substitution established that the 2 Zn hexamer is a calcium binding protein (Sudmeier et al., 1981). A variety of physico-biochemical studies have since shown that one calcium ion binds at the centre of each hexamer in a cage formed by the six B13 Glu carboxylates with a dissociation constant of 80 microM (Sudmeier et al., 1981 ; Storm and Dunn, 1985 ; Alameda et al., 1985 ; Kaarsholm and Dunn, 1987 ; Dunn et al., 1987 ; Palmieri et al. 1988 ; Coffmann and Dunn, 1988). Substitution of a calcium ion into the metal-free B13 Glu cavity disrupts the intra-dimer Glu-Glu hydrogen-bonding interaction, and the single ion has to move rapidly between the three symmetry related sites in order to coordinate the covalently seperate molecules. In this respect the calcium binding site of the 2 Zn hexamer is quite unlike any other known protein structure (Hill et al., 1991). The presence of calcium at the centre of the hexamer has a stabilising effect by eliminating the coulombic effects whih would otherwise arise from ionisation of the B13 carboxyls in its absence. It has been shown that pancreatic storage granules contain high concentrations of Ca2+ in addition to Zn2+ (Herman et al., 1973), and their physiological role is probably to facilitate hexamer formation from proinsulin and stabilization of the complex during storage.
Several mutant insulins have been crystallised with recipies that differ from those traditionally used, probably owing to their altered stability and solubility (Xiao et al., 1992).

|
|
Raj Gill, John Walshaw, John Kenney
Last updated 14th April '97