

|
Fiberous proteins, such as collagen and intermediate filaments, are extended structures hundreds of nm long. They form parallel arrays to make larger, stronger quaternary structures called fibers. An example of a structure composed of fibers is the extracellular matrix. This protein and polysaccharide matrix acts as a universal biological glue, as well as forming specialized structures including tendons, bone, cartilage and basal laminae. The matrix consists of fibrous proteins in a hydrated polysaccharide gel. The major class of extracellular proteins consists of the collagens; in fact collagens constitute a quarter of a mammal's entire protein content.
A collagen molecule consists of three polypeptide chains arranged in a parallel triple-helix. The individual polypeptide chains are called alpha-chains, and although each is helical they only have this conformation when associated with two other alpha chains; they should not be confused with alpha-helices. There are at least 9 distinct alpha chains; most are approximately 1000 amino acids in length. This results in a diameter of 14 Å and a length of about 300nm for a single collagen molecule.
The helical conformation of each chain is dependent on the fact that every third residue is a Gly, and that the sequence is rich in Pro. A larger side chain than Gly would prevent the close contact of the three chains. About half of the Pro side chains are hydroxylated; the resulting residue Hydroxproline is referred to as "Hyp". The structure is stabilised by hydrogen bonds between the backbone amide of a Gly residue and the backbone carbonyl of residue X, where the sequence is represented by a repeat of -Gly-X-Y-. Collagen I, which constitutes 90% of the collagen in a mammal, consists of a heterotrimer where 2 of the alpha-chains are identical.
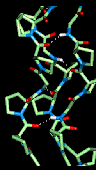 Here is a model
collagen structure. This consists of the repeated
sequence Gly-Pro-Pro in each of the 3 alpha-chains, subjected to an energy
minimization procedure. Hydrogen bonds between the backbone atoms of the
3 alpha-chains are indicated.
Here is a model
collagen structure. This consists of the repeated
sequence Gly-Pro-Pro in each of the 3 alpha-chains, subjected to an energy
minimization procedure. Hydrogen bonds between the backbone atoms of the
3 alpha-chains are indicated.
![]() This model
can be seen with RasMol by clicking here
1clg (62Kb)
[Bbk|BNL|ExP|Waw|Hal].
This model
can be seen with RasMol by clicking here
1clg (62Kb)
[Bbk|BNL|ExP|Waw|Hal].
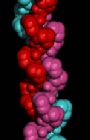 Here is a space-filling
model of the triple-helix. To obtain a similar rendition with RasMol,
colour the structure by chain, and use the space-filling option.
Here is a space-filling
model of the triple-helix. To obtain a similar rendition with RasMol,
colour the structure by chain, and use the space-filling option.
The alpha-chains are initially synthesized in the form of precursors called pro-alpha-chains, which have globular sequences called extension peptides at each end. The C-terminal extension peptides (approximately 300 residues long) of three chains associate, forming disulphide cross-links, and the triple helix forms in a zipper-fashion giving procollagen. Collagen results from the cleavage of the extension peptide domains.
Collagen type I forms collagen fibrils. Collagen molecules are arranged head-to-tail, with a 35nm gap between each, in a staggered bundle. All the molecules point in the same direction. The -OH groups of hydroxyproline are involved in hydrogen bonds between chains, while interactions between other side chains are thought to be important in formation of fibrils from a number of individual molecules. Charged and uncharged residues are found to be periodically clustered along the sequence of collagen I every 234 residues, which is equivalent to 67 nm; while the gap between staggered molecules in a fibril is the same distance. This suggests that the collagen molecules are aligned such that the maximum electrostatic and hydrophobic interactions occur between different molecules.
The diagram here indicates interactions between alpha-chains in two different molecules. Again this is taken from a model structure, of the Smith collagen microfibril arrangement in which 5 triple helices are involved in a left-handed superhelical arrangement which is postulated to be repeated throughout the fibril. Only two of the five collagen molecules are shown here. All the alpha-chains consist of a repeated Gly-Pro-Hyp sequence. Three Hyp residues are highlighted forming hydrogen bonds between individual collagen molecules, rather than within them.
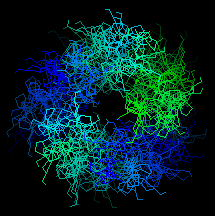
![]() Here is an
end-on view of the superheilx. Click here
4clg
(329Kb)[Bbk|BNL|ExP|Waw|Hal]
to see the whole structure. Colour by chains.
Here is an
end-on view of the superheilx. Click here
4clg
(329Kb)[Bbk|BNL|ExP|Waw|Hal]
to see the whole structure. Colour by chains.
Silk fibroin, tropomyosin and keratin are other fibrous proteins which are rich in glycine. Details of these can be found under the Glycine Group's section on Glycine in Structure.
![]() tropomyosin
structure 2tma (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
tropomyosin
structure 2tma (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
Intermediate filaments are 7-11nm in diameter, and are therefore intermediate in size between the other major cytoplasmic filaments, namely microtubules and microfilaments (which are aggregations of many globular subunits, rather than fibrous proteins; see below).
There are five major types:
Note that due to their long, fibrous nature, crystallography is not a very successful approach to determining the structure of fibrous proteins.
The morphology, movement and division of the cell depend on the cytoskeleton. It is composed primarily of filamentous structures. Two major types are microfilaments (also known as actin filaments) and microtubules. Both of these structures are aggregations of globular protein subunits into filaments or tubules upto several microns long. These rapidly rearrange by predominately polymerising at one end and depolymerising at the other.
At least six different types of actin are synthesized by vertebrate cells. However, their amino acid sequences are highly conserved and they have similar properties. Globular, or G actin is stabilized by one tightly bound calcium ion and is associated with one molecule of ATP. The G actin polymerizes to form filamentous or F actin. This involves hydrolysis of the terminal phosphate of the ATP. Actin filaments consist of two strands of polymerized actin monomers, twisted into a helix with 13.5 molecules per turn.
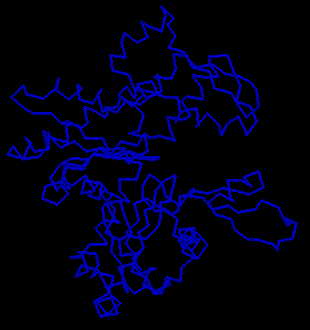
![]() Examine the crystal structure of actin
1atn
(434Kb)[Bbk|BNL|ExP|Waw|Hal]
The actin molecule is complexed with deoxyribonuclease I; actin is the larger
of the two (RasMol command: restrict *A). Consider
how the shape of this molecule might relate to its ability to polymerize.
Examine the crystal structure of actin
1atn
(434Kb)[Bbk|BNL|ExP|Waw|Hal]
The actin molecule is complexed with deoxyribonuclease I; actin is the larger
of the two (RasMol command: restrict *A). Consider
how the shape of this molecule might relate to its ability to polymerize.
The globular monomeric form is called G-actin, and has a bound calcium ion and a bound ATP molecule, which is hydrolyzed when the monomers polymerize to form F-actin . Electron micrographs indicate the parallel double-helical form of these filaments. Each turn of the helix has 13.5 monomers, and is 360Å long. The diameter is approximately 70Å. The structure is indicated below.
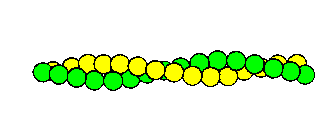
Actin filament. Each sphere represents a globular actin monomer.
Actin filaments are the major component of thin filaments of muscle tissue. Each group of seven monomers in one strand of the helix has a bound molecule of tropomyosin, and one bound molecule of each of three troponin peptides: Tn-T, Tn-I and Tn-C:
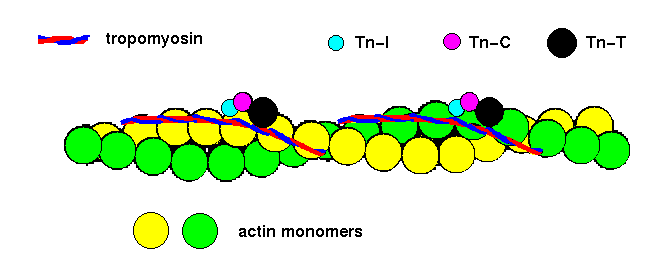
![]() tropomyosin
2tma (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
tropomyosin
2tma (55Kb)
[Bbk|BNL|ExP|Waw|Hal]
Tn-C 5tnc
(132Kb)[Bbk|BNL|ExP|Waw|Hal]
Muscle fibres and their proteins are examined in more detail in a later section of the course.
Microtubules are composed of dimers of the protein tubulin. The heterodimer consists of one alpha-tubulin and one beta-tubulin subunit (about 40 Å in size). The two subunits are homologous. These dimers are aligned head-to-tail in rows called protofilaments. A variable number of protofilaments (from 11 to 17) form the hollow tube that is the microtubule. In vivo, most microtubules contain thirteen protofilaments. Each protofilament is staggered about 9Å with respect to its neighbours, so that the tubulin subunits describe helicies. The diameter of the microtubule is approximately 240Å.
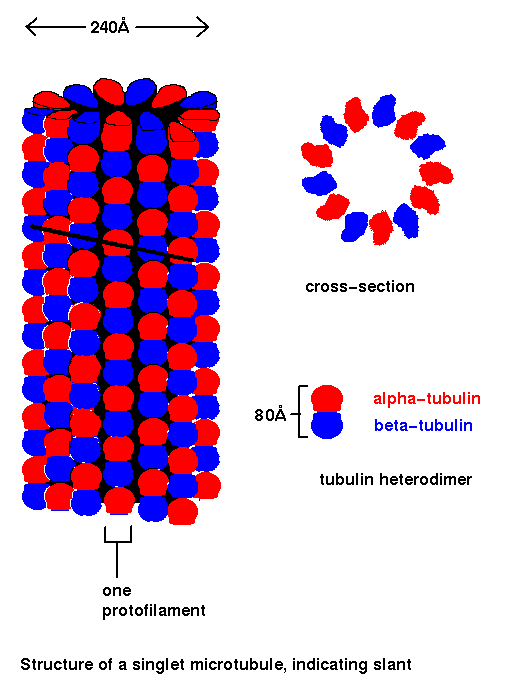
A doublet microtubule consists of 23 protofilaments in a figure-of-eight arrangement. A ring of nine doublet microtubules, with two singlet microtubules at the centre, forms the basis of cilia and flagella .
Viruses have a genome of nucleic acids enclosed by a protective shell of protein. In many cases, this nucleocapsid is enveloped by a lipid bilayer and other proteins. Though viral genomes code for only a few (typically less than a dozen) different proteins, many copies (usually hundreds) of these proteins are required to form the nucleocapsid. In order to form the nucleocapsid so that similar proteins don't have to make different (or very different) contacts, the proteins are symmetrically arranged. The nucleocapsid must be closed (to contain the genome) and posses a high symmetry (to allow the use of many copies of the same protein). Cylinders exhibitng helical symmetry and spherical structures exhibiting icosahedral symmetry satisfy these requirements. Indeed, many viruses are helical, icosahedral or a combination of the two.
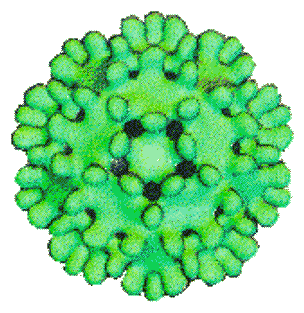
The icosahedral nucleocapsid of duck hepatitis B virus
For a thorough description of virus structure, assembly and symmetry (including an extensive reference list), you should study Virus Coat Protein Structures by PPS Consultant Gail I. Schuman, at the Biology Department, Brookhaven National Laboratories, U.S.
Further material on the structure of viral protein coats is to be found in
the Institute for Molecular Virology server at Wisconsin-Madison. In particular,
there is a section on
visualization of virus
proteins .
(Institute for Molecular Virology
Home Page )
At the Wageningen Agricultural University , Netherlands, examine the material
by Sander Meijer and Anton Feenstra on the various types of symmetry
exhibited in a
typical icosahedral virus (CCMV).
(WAU Transitorium homepage)
To look through a large list of WWW virology servers look at the web pages of Garry Lab.

|
John Kenney, John Walshaw
Last updated 14th April '97