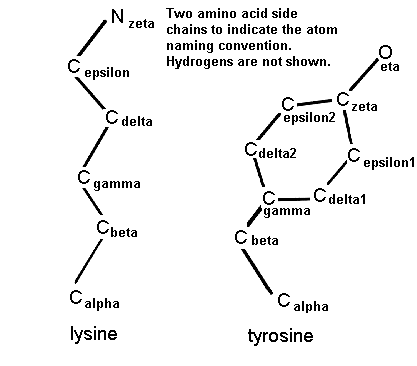
The side chain atoms of amino acids are named in the Greek alphabet according to this scheme.

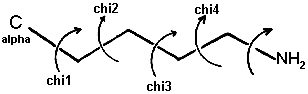
The side chain torsion angles are named chi1, chi2, chi3, etc., as shown below for lysine.
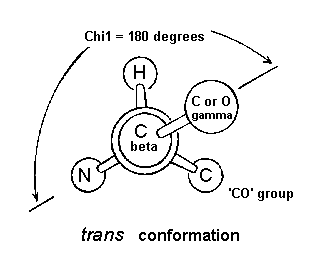
The chi1 angle is subject to certain restrictions which arise from steric hindrance between the gamma side chain atom(s) and the main chain. The different conformations of the side chain as a function of chi1 are refered to as gauche(+), trans and gauche(-). These are indicated in the diagrams below in which the amino acid is viewed along the Cbeta-Calpha bond.
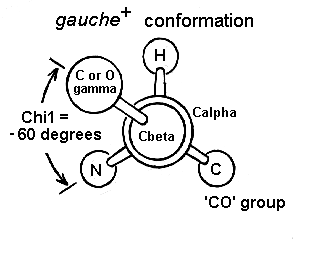
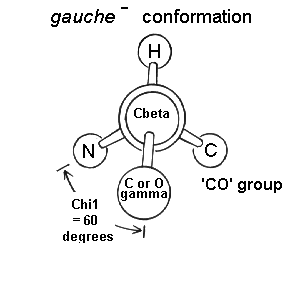
The most abundant conformation is gauche(+) in which the gamma side chain atom is opposite to the residue's main chain carbonyl group when viewed along the Cbeta-Calpha bond.
The second most abundant conformation is trans in which the side chain gamma atom is opposite the main chain nitrogen.
The least abundant conformation is gauche(-) which occurs when the side chain is opposite the hydrogen substituent on the Calpha atom. This conformation is unstable because the gamma atom is in close contact with the main chain CO and NH groups. The gauche(-) conformation is occasionally adopted by serine or threonine residues in a helix where the steric hindrance is offset by a hydrogen bond between the gamma oxygen atom and the main chain.
With most amino acids the gauche(+) and trans conformations are adopted with similar abundances although the gauche(+) conformation tends to dominate.
Aliphatic amino acids which are bifurcated at Cbeta, ie valine and isoleucine, do not adopt the trans conformation very often as this involves one of the Cgamma atoms being in the unfavourable gauche(-) 'position'.
In general, side chains tend to adopt the same three torsion angles (+/-60 and 180 degrees) about chi2 since these correspond to staggered conformations. However, for residues with an sp2 hydridised gamma atom such as Phe, Tyr, etc., chi2 rarely equals 180 degrees because this would involve an eclipsed conformation. For these side chains the chi2 angle is usually close to +/-90 degrees as this minimises close contacts. For residues such as Asp and Asn the chi2 angles are strongly influenced by the hydrogen bonding capacity of the side chain and its environment. Consequently, these residues adopt a wide range of chi2 angles.
`Principles of protein structure', G.E.Shultz and R.H.Schirmer, Springer-Verlag, New York.
`Structure and function of proteins', R.E.Dickerson and I.Geis, Harper and Row, London.
`The Nature of the chemical bond', L.Pauling, Cornell University Press, New York.
`Solvent induced distortion and curvature of alpha-helices', T.Blundell, D.Barlow, N.Borkakoti and J.Thornton. Nature (1983) 306, 281-283.
`Helix geometry in proteins', D.Barlow and J.Thornton. J.Molec.Biol. (1988) 201, 601-619.
`Conformation of beta-hairpins in protein structures. A systematic classification with applications to modelling by homology, electron density fitting and protein engineering', B.L.Sibanda and J.M.Thornton. J.Molec.Biol. (1989) 206, 759-777.
`Stereochemical quality of protein structures', A.L.Morris, M.W.MacArthur, E.G.Hutchinson and J.M.Thornton. Proteins (1992) 12, 345-364.
 Back to the Top
Back to the Top