
SH3 (Src homology region 3) domains are found in many signal transduction proteins including Src, Abl, Fyn, Lyn and Grb-2. These non-catalytic domains bind to peptides and proteins with a proline containing sequence. The sequences fall into two classes, class I and class II which bind I opposite (N to C) orientations. All have a PXXP motiff, with class I having PXXXPXXP or RXXPXXP and class II XPXXPXR(K).(4) The proline-rich sequences have been shown to adopt a left-handed poly-proline type II helix(5). The 3-dimensional structures of many of these are known, some with peptides bound. The structure of cSRC with a bound peptide is shown below. Of the six residues within 5 angstroms of a pepetide proline, 3 are aromatic and anther is also a proline.
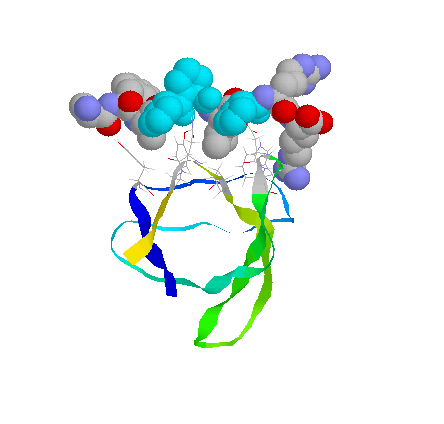
cSRC SH3 domain with Peptide
Peptide is in spacefill, residues within 5A are wireframe cSRC in ribbons.
From PDB# 1PRM, rendered with RASMOLE
WW domains are a recently found proline containing sequence in signal transduction proteins. It was named for the two conserved tryptophans(W) and also has two conserved prolines at the N and C termini. WW domains have the concensus sequence of:
p = polar and h = hydrophobic
WW domains are found in YAP(Yess associated protein), and dystrophin, the protein associated with muscular dystrophy .