Back to main Molecular Forces index
Back to main PPS course Index

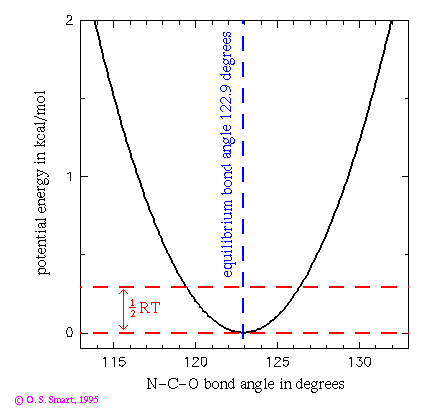
This graph shows the potential energy for a N-C-O bond angle using a
harmonic potential with the parameters given
in the AMBER potential energy
function. Such a term would partly control the angular vibrations
of the carbonyl oxygen on the main chain of a polypeptide chain.
The bond angle constant 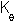 is
80 kcal/(mol.degrees^2) and the equilibrium bond angle
(
is
80 kcal/(mol.degrees^2) and the equilibrium bond angle
(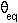 ) is 122.9 degrees.
The dashed line indicates an energy of 0.29 kcal/mol which is equal
to 1/2RT at a temperature of 300K. This is the energy that an
individual degree of freedom can expect at this temperature and
indicates that a this bond could be expect to be undergoing
vibrations of the order of 4 degrees at room temperature.
If we assumes that the N and C atoms remain mostly static
then this would entail motion in this degree of freedom of the order
of 0.09 Å
for the oxygen atom (as the C-O bond is 1.229 Å long).
It is interesting to compare this to a motion of
approximately 0.03 Å amplitude to vibrations of the C=O bond
(link to graph of this motion).
(This is not quite the whole story as there would be another bond
angle term for the atoms C_alpha-C-O also restricting the motion).
) is 122.9 degrees.
The dashed line indicates an energy of 0.29 kcal/mol which is equal
to 1/2RT at a temperature of 300K. This is the energy that an
individual degree of freedom can expect at this temperature and
indicates that a this bond could be expect to be undergoing
vibrations of the order of 4 degrees at room temperature.
If we assumes that the N and C atoms remain mostly static
then this would entail motion in this degree of freedom of the order
of 0.09 Å
for the oxygen atom (as the C-O bond is 1.229 Å long).
It is interesting to compare this to a motion of
approximately 0.03 Å amplitude to vibrations of the C=O bond
(link to graph of this motion).
(This is not quite the whole story as there would be another bond
angle term for the atoms C_alpha-C-O also restricting the motion).