 All-Beta Topologies
All-Beta Topologies
![]() Index to Course Material
Index to Course Material
![]() Index to Section 10
Index to Section 10
![]() All Alpha Folds
All Alpha Folds
![]() Alpha/Beta Folds
Alpha/Beta Folds
You should also browse through the Mainly Beta Class of the CATH Protein Structure Classification Database at University College, London. This lists 7 different architectures.
You should also be aware of alternative classifications, such as the Structural Classification of Proteins database (Alexey G. Murzin, Steven E. Brenner, Tim J.P. Hubbard, and Cyrus Chothia). In all, fifty-two categories of all beta folds are listed. A number of the entries have links to diagrams by Manuel Peitsch.
With MAGE installed, study this Kinemage on Alpha Domain Structures, which accompanies the Branden and Tooze book.
 Click here for a diagram
of this beta sheet arrangement in
the lipocalin intestinal fatty acid-binding protein.
Click here for a diagram
of this beta sheet arrangement in
the lipocalin intestinal fatty acid-binding protein.
![]() 1ifb (95Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
1ifb (95Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
This is just one member of the Lipocalin family, which bind small molecules between the sheets of the sandwich.
![]() 1stp (87Kb)
[Bbk|BNL|ExP|Waw|Hal]
See also
the diagrams of porin in the section on
membrane proteins in the previous chapter. Note that some structures are
intermediate between the extreme barrel and sandwich arrangements.
1stp (87Kb)
[Bbk|BNL|ExP|Waw|Hal]
See also
the diagrams of porin in the section on
membrane proteins in the previous chapter. Note that some structures are
intermediate between the extreme barrel and sandwich arrangements.

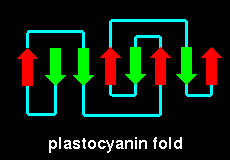 plastocyanin (21Kb GIF).
plastocyanin (21Kb GIF).
![]() 2plt (71Kb)
[Bbk|BNL|ExP|Waw|Hal]
Here is the crystal
structure. Notice that this has a mixed sheet- there are two parallel
pairs of strands. This SCRIPT renders the molecule
as in the diagram.
2plt (71Kb)
[Bbk|BNL|ExP|Waw|Hal]
Here is the crystal
structure. Notice that this has a mixed sheet- there are two parallel
pairs of strands. This SCRIPT renders the molecule
as in the diagram.
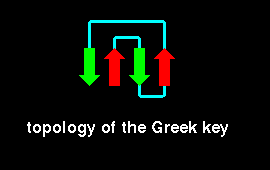
![]() This can be seen by examining the crystal structure of gamma-crystallin.
2gcr (136Kb)
[Bbk|BNL|ExP|Waw|Hal]
This SCRIPT displays only the N-terminal domain, and
colours the two sheets as in the diagram above (white and red). To distinguish the
two Greek keys, type the following:
This can be seen by examining the crystal structure of gamma-crystallin.
2gcr (136Kb)
[Bbk|BNL|ExP|Waw|Hal]
This SCRIPT displays only the N-terminal domain, and
colours the two sheets as in the diagram above (white and red). To distinguish the
two Greek keys, type the following:
select 1-39 colour blue select 40-80 colour [90,90,70]Sequence homology has been found between the two Greek key motifs within each domain, and also between the two domains themselves. The latter homology is higher than the former; this implies that the structure evolved from a single Greek key fold by means of a gene duplication to produce a domain of two Greek keys, followed by a second duplication resulting in two similar domains. This is supported by the fact that in some crystallins each Greek key motif is coded by a different exon, with introns between them.
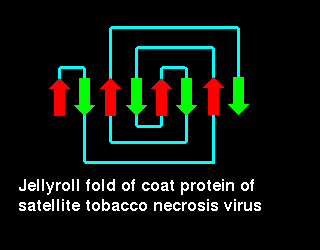 Click here for a diagram
illustrating this fold in the coat protein of
satellite tobacco necrosis virus (21Kb GIF).
Click here for a diagram
illustrating this fold in the coat protein of
satellite tobacco necrosis virus (21Kb GIF).
![]() 2stv (158Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT to render as in
the diagram
2stv (158Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT to render as in
the diagram
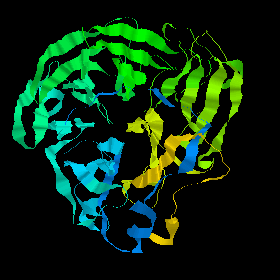
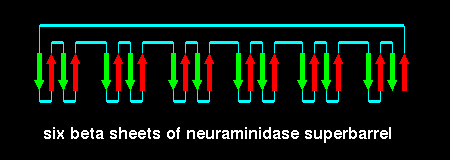
This fold is called a beta propellor; there are six 'blades' in neuraminidase.
![]() Here are two other examples - how many blades are there in each propellor?:
Here are two other examples - how many blades are there in each propellor?:
![]() Examine the crystal
structure of erythrina trypsin inhibitor 1tie (114Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
Examine the crystal
structure of erythrina trypsin inhibitor 1tie (114Kb)
[Bbk|BNL|ExP|Waw|Hal]
... SCRIPT
Last updated 26th Jun '96